|
This is the basis for the battery experiments we detail elsewhere on our blog. This review briefly describes the components of the sodium battery, including the anode, cathode, electrolyte, binder, and separator, and the sources of sodium raw material is the most important in material synthesis or installation. 
Any two, conducting materials can form an electrochemical cell, provided they have different standard potentials. The main components anode and cathode have significant effect on the sodium battery performance. This term refers to its ability to transmit, or receive electrons. 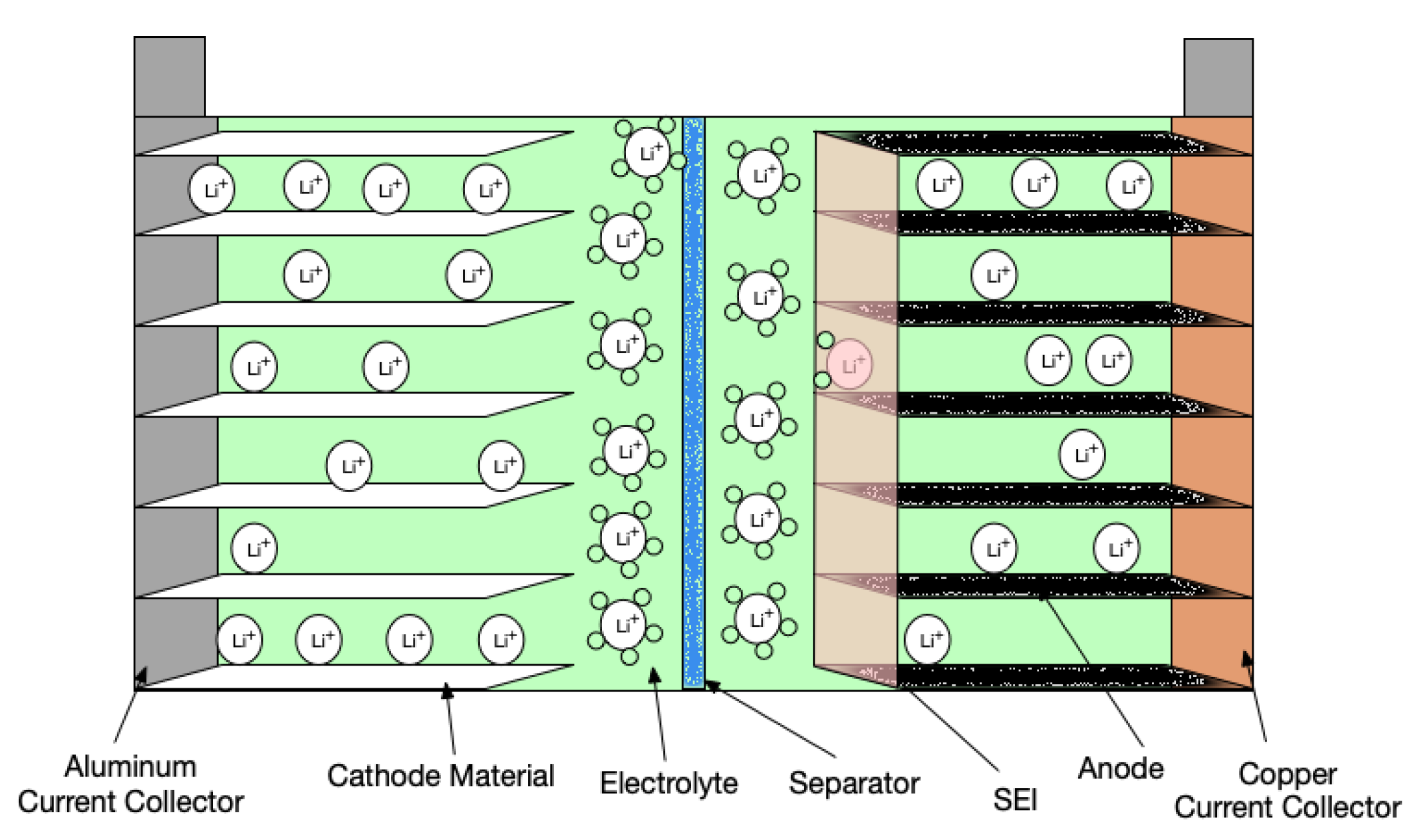
A second chemical reaction occurs at the cathode, which prepares it to receive those electrons via an external circuit.Įach of these reactions has what we call a particular standard potential.They can be operated between 7 and about +50 ☌. This is when hydrogen accumulates at the cathode, reducing the battery's effectiveness. Primary batteries become polarized with use. Warmer temperatures can also lower the performance of the battery, by speeding up the side chemical reactions. Most zinccarbon batteries are cathode limited, as the anode also acts as the battery can. The rate of side reactions can be slowed by lowering temperature. Both the carbon and the manganese dioxide should contain very little active impurities that can migrate to the zinc surface. Battery chemistry begins as the anode reacts with the electrolyte, to produce electrons which accumulate there. The cathode mix contains up to 75 by volume of liquid.There is also a third substance, the electrolyte. These are usually different metals, or other substances or compounds.We call the negative electrode the anode, and the positive one the cathode. This process begins at one of the electrodes, where a chemical reaction releases electrons to flow to the other one: Ions simultaneously flow between the electrodes restoring the balance.Ĭlearly, a battery needs to get its energy from somewhere before it can perform. The flow of electrons alters the battery chemistry.This energizes any electrical device in the external circuit. This flow of electrons constitutes an electric current.Chemical reactions inside batteries invoke the flow of electrons between the terminals via an external circuit.Batteries are physical devices with electrodes that store chemical energy, and transform this into electrical energy.How Battery Chemistry Works: A Brief Overview 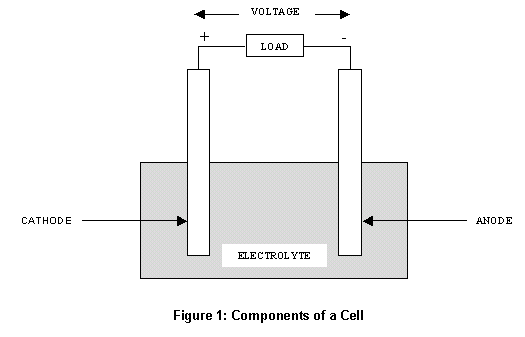
Read on to discover which substances are involved in batteries, and how these react together. We delve into battery chemistry in this article. This is with particular reference to ‘their properties and reactions, and the use of such reactions to form new substances’. Chemistry, Oxford Languages informs us concerns the substances that make up matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
